
Non-metals have high ionization energies and high electron affinities, so they can gain electrons easily, but lose them with much more effort, causing them to be classified as anions. Unlike metals, Non-metals are often very poor conductors of heat and electricity.Non-metals often present with lower densities.Non-metals have low melting and boiling points.Generally, non-metals are not shiny or lustrous.Like bromine which might be found as a liquid. The majority of metals are found in a gaseous state, but you can find some in a solid or liquid state.The non-bonding helium, neon, radon, argon, xenon, krypton, and oganesson, also known as the noble gasses, make up a chunk of the non-metals. While there are only seventeen non-metals on the periodic table a few common examples include oxygen and nitrogen which account for most of the air that we breathe, along with a few other gases like neon or the chemical compound carbon dioxide. This picture does not include Hydrogen which is also a non-metal. Living things are made up of almost entirely non-metals like carbon or hydrogen. Many of these elements have biological roles. Non-Metals also include hydrogen which lies to the left of the metalloids. On the periodic table, non-metals lie to the right of the zigzag line that runs between the elements boron, silicon, arsenic, tellurium, and astatine. Non-Metals account for a small portion of elements on the periodic table. When these metals bond with other non-metals this is known as ionic bonding where a transfer of electrons occurs from one species to another. Metals that follow the transition metals can lose their s orbital and p orbital electrons and form charges that are the same as their group number or they can lose their p orbital electrons and keep their two s orbital electrons metals like tin and lead in group 4A can form either +4 or +2 charges. The metals in group 1A like Lithium and Sodium have a charge of +1, the metals in group 2A such as magnesium and calcium have a +2 charge. 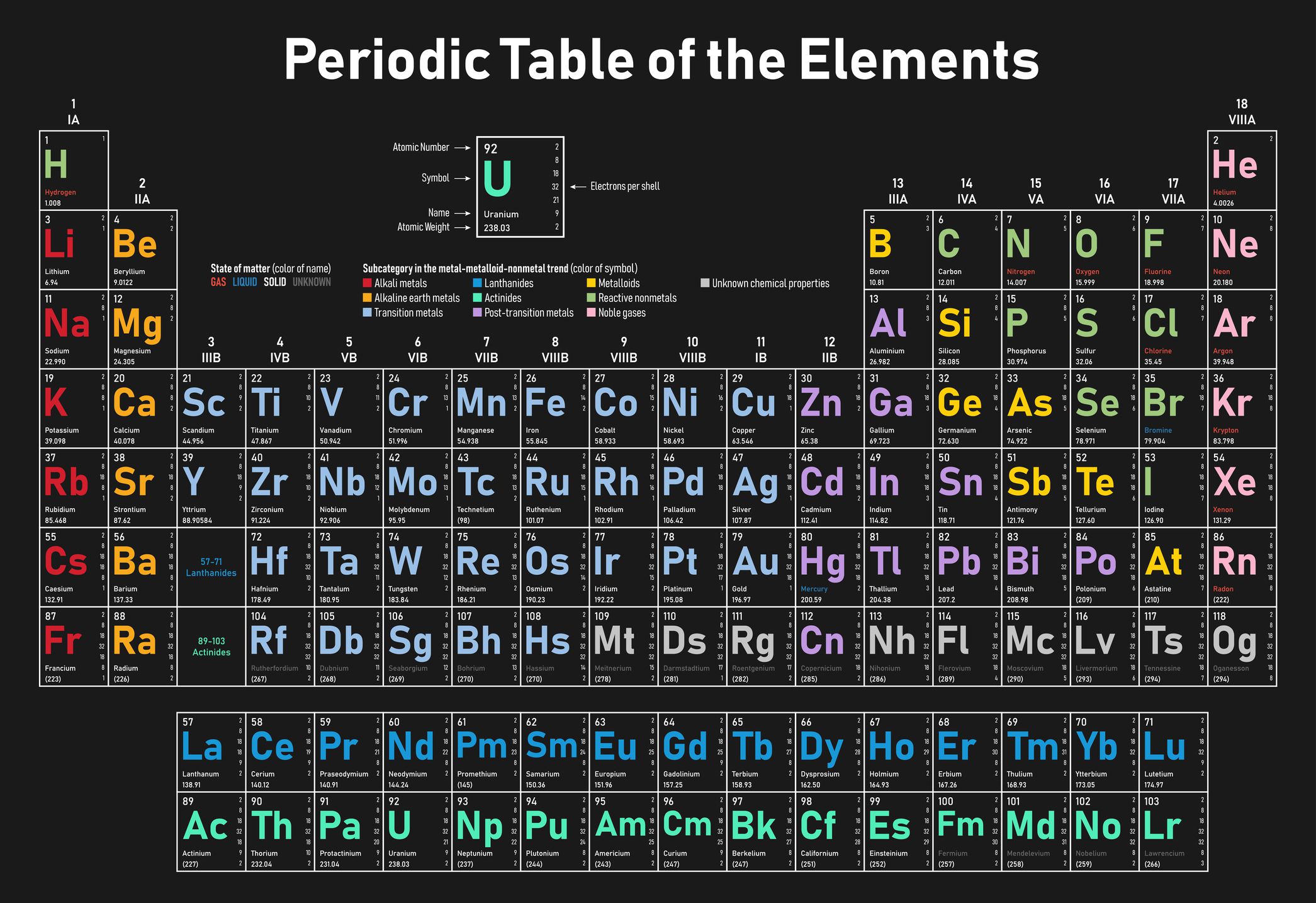
The main group metals usually form charges that are the same as their group number. Since the metals lie to the left of the periodic table they often have low ionization energies and low electron affinities, meaning they give away electrons relatively easy causing metals to become cations.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
/PeriodicTableoftheElements-5c3648e546e0fb0001ba3a0a.jpg)
 RSS Feed
RSS Feed
